Bimeda® Announces Re-introduction of Ovacyst® and Prostamate®
Oakbrook Terrace, Illinois- March 7, 2022- Bimeda® Animal Health has announced the expansion of its US product portfolio with the re-introduction of OvaCyst® (gonadorelin) and ProstaMate® (dinoprost tromethamine injection).
These recognizable brands (formerly marketed by Bayer HealthCare LLC Animal Health Division following their acquisition of Teva Pharmaceutical Industries Ltd. in 2013) provide veterinarians and producers familiar molecules that they have relied on for years at a cost-effective price.
"Reproduction is the single-most important factor for profitable cattle production." said Dr. Chris Thomsen, Senior Technical Services Veterinarian, Bimeda Inc. "Utilization of reproductive hormones for estrus synchronization can reduce or eliminate the need for bulls, decrease time and labor costs associated with heat detection, and shorten the calving interval. Reproductive hormones provide veterinarians and producers effective tools to maximize reproductive efficiency which will positively impact an operation's profitability. Bimeda is excited to return OvaCyst and ProstaMate to the reproductive hormone market."
About Bimeda's New Reproductive Management Products:
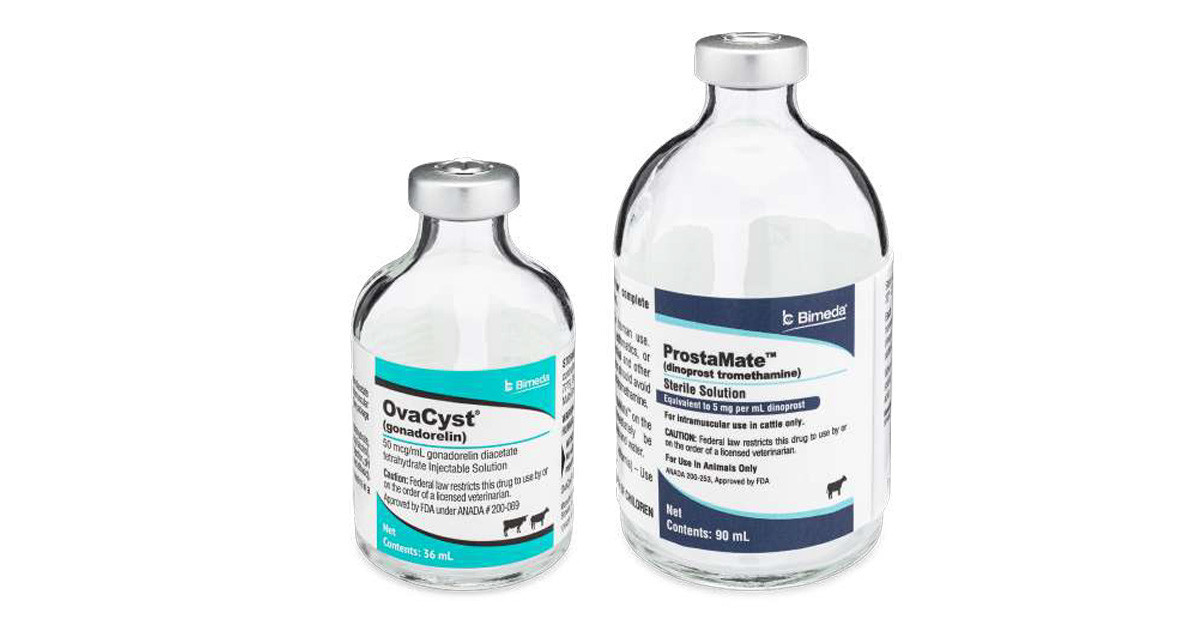
OvaCyst is a sterile solution containing 43 mcg/mL of gonadorelin (GnRH) as gonadorelin diacetate tetrahydrate and is indicated for treatment of cystic ovaries in dairy cattle and reproductive synchrony in beef and dairy cattle.
ProstaMate is a sterile solution that contains 5 mg/mL of dinoprost as dinoprost tromethamine—a naturally
occurring prostaglandin F2-alpha. It is indicated for estrus synchronization, treatment of unobserved estrus,
abortion and chronic endometritis in cattle. Additionally, ProstaMate is approved for use with gonadorelin
injection for Fixed-Time Artificial Insemination (FTAI) and EAZI-BREEDTM CIDR® (progesterone intravaginal cattle insert).
OvaCyst and ProstaMate are both currently available through Bimeda's distributor partners. OvaCyst is available in a 36 mL vial and is approved for intravenous (IV) or intramuscular (IM) administration with a recommended dosage in cattle of 100 mcg (2 mL). ProstaMate is approved for IM administration only and is available in a 90 mL vial with a recommended dosage in cattle of 25 mg (5 mL).
For more information about ProstaMate and OvaCyst, visit our product page or contact a Bimeda Sales Representative.